Everything You Ever Wanted To Know About Lithium Ion Batteries
Although initial research and pioneering work for the lithium battery began as early as the year 1912, it was not commercially available until 1972. More research and advancements leading to rechargeable lithium-ion batteries continued into the 1980s. These early attempts were based on metallic lithium and offered very high energy density. Unfortunately, the highly instable properties of the lithium metal, particularly while being charged, posed too high of a risk for manufacturers, so research and development slowed down substantially during that period. Although the scientific reasoning behind this instability would be an entire research paper in and of itself, the summary is that lithium cells had the potential of a “thermal run-away.” In other words, the temperature would rapidly rise to its melting point, which in turn caused a violent, and sometimes explosive, reaction. In fact, in 1991, a large number of rechargeable lithium-ion batteries had to be recalled after a cellular phone released hot gases and inflicted severe burns to a man's face while in use.
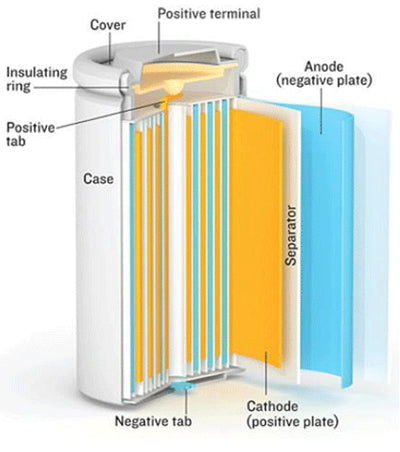
As a result of this instability and its potential safety hazards, manufacturers shifted their research from lithium metal to a non-metallic lithium battery using lithium ions instead. The first commercially-available lithium-ion battery was manufactured by Sony. The downside to using lithium ions rather than the lithium metal is that lithium ions are a bit lower in energy density. However, this is grossly outweighed by the fact that the lithium ion battery system is much more stable, and therefore, safer than its predecessor. Today, lithium-ion batteries are one of the most widely used battery systems available. With over two billion batteries produced annually, lithium-ion technology is one of the most successful battery chemistries on the market. They are also safe batteries, provided that certain important safety measures are met.
Aside from the fact that lithium-ion cells with cobalt cathodes hold twice the energy as do nickel-based batteries, and four-times that of lead acid battery systems, there are other big advantages to them as well. The lithium-ion battery system requires much less maintenance than its counterparts. Moreover, they don’t have memory and do not require scheduled cycling to ensure a long life. Furthermore, Lithium-Ion batteries do not have the issue of “sulfation” that results when lead acid batteries are stored without periodic charges to top the power off. Lithium-ion batteries are also much more environmentally-friendly, as they have relatively low self-discharge.
Unfortunately, as with many technological advancements, the benefits of lithium-ion batteries do not come without a price. There have been several recalls in the past due to unsafe and potentially-hazardous batteries. The highest profile case came when Dell and Apple were forced to recall over 6 million lithium-ion battery packs that were being used in select laptops, and that were manufactured by Sony. According to Sony, on very rare occasions, foreign microscopic metal particles come into contact with other areas of the battery cell, which in turn, leads to a short circuit within the cell. Although battery manufacturers strive to minimize the presence of metallic particles, complex assembly techniques make the elimination of all metallic dust nearly impossible.
On a macro level, there are essentially two forms of lithium-ion systems: Manganese (Spinel), which uses the product code: IMR, and Cobalt, which uses the product code: ICR. Since cobalt systems have a higher runtime level, often-used and energy intensive products, such as laptops and mobile phones, generally use cobalt over manganese systems. Manganese, being a newer technology, offers better thermal stability and can sustain temperatures of up to 482°F (250°C) without compromising stability. The downside to spinel/manganese is that it contains lower energy density, providing approximately 50% the capacity of cobalt systems. To find a happy median, manufacturers generally mix the two systems to maximize the strengths of each, while limiting the downside.
Some lithium-ion batteries are high-drain, which means that the cell can handle high output currents. Cobalt systems are generally limited to two times the charge capacity in output drain. As an example, a 750mAh 14500 cobalt lithium-ion battery cell would be limited to two times 750mA or 1.5A. It is specifically for this reason that cells of this sort must be internally protected. It has the lowest drain capability and the highest volatility or flammability in composition. Maganese or Spinel systems, on the other hand, generally have an 8C limit. That is, it is limited to eight times the charge capacity. For example, a manganese lithium-ion battery 600mAh 14500 can withstand 4.8A. Due to the fact that Spinel systems are much less volatile and maintain higher drain limits, they are typically not internally protected. External protection, however, for manganese lithium-ion batteries is recommended, as they too are susceptible to burning, albeit a much lesser chance.

Generally, Spinel systems are used for variable voltage and variable wattage devices. This is due to the fact that variable voltage and wattage devices require what are known as boosters to allow for the rapid adjustment of voltage or wattage. The adjustment essentially means that the device is jumping from higher to lower currents and vice versa, which requires high drain. Lithium-ion batteries are generally safe, and problems with them are very rare. Manufacturers of these batteries have various safety measures in place to drastically reduce the potential for any malfunction. However, it is critical to understand that the safety precautions are only useful if the harm is coming from outside the battery itself, such as a defective charger. If the internals of the battery is compromised, such as contamination caused by foreign microscopic metal particles, the safety features of the battery will be ineffective.
One big safety concern is when the user attempts to charge the lithium-ion battery in cold temperatures below 32°F (0°C)—the freezing point of water. Lithium-ion batteries absolutely cannot and should not be charged below this temperature. Inasmuch as the battery may seem to be charging, “plating” is actually occurring on the anode, which makes the battery substantially more vulnerable to failure.
Another big safety concern involves defective chargers and/or static electricity damaging the battery’s protection circuit. This type of damage can potentially fuse the solid-state switch to the “on” position without the user knowing that this is occurring Moreover, a third potential hazard may arise from off-brand batteries being purchased to replace the batteries that originally came with the device. Non-brand name batteries advertise as being an equivalent battery, but with a lower price; however, the majority of these batteries do not afford the same level of safety as the brand-name counterpart. It is highly recommended that replacement batteries either be the same brand as that which came with the device. Otherwise, you should purchase only well-known and trusted brand names. Equally important, you must always make sure to use the proper charger for the specific battery you are charging. Use of the wrong charger can be extremely dangerous. So long as the proper precautions are met, lithium-ion batteries are perfectly safe to use.










